OCREVUS Pregnancy Registry
What is the OCREVUS Pregnancy Registry?
The OCREVUS Pregnancy Registry is a global registry for women with MS who are pregnant and have or have not received OCREVUS® (ocrelizumab). The purpose of this pregnancy registry is to learn more about the health of pregnant women who have or have not received ocrelizumab and the health of their babies. This will be done by monitoring participants throughout their pregnancy, followed by monitoring the health of their babies until they are at least 12 months old.
Because of this registry, pregnant women who are exposed to ocrelizumab in the future may have more information about how their pregnancies and babies may or may not be affected.
Can I take part in this registry?
You may be able to take part in this registry if you are 18 years or older and pregnant, have MS and:
- have received ocrelizumab at any time since 6 months before your last menstrual period, or
- have not received ocrelizumab or any other MS disease-modifying therapy, except glatiramer acetate, at any time since 6 months before your last menstrual period.
What will I have to do?
To take part in the registry, you will first be asked to give informed consent during a telephone interview with a registry representative. This means that you will receive information about the registry and what it will involve, have a chance to ask any questions, and decide if you want to take part.
If you decide to participate and give your informed consent, then a registry representative will:
- collect some information about you and your pregnancy via telephone
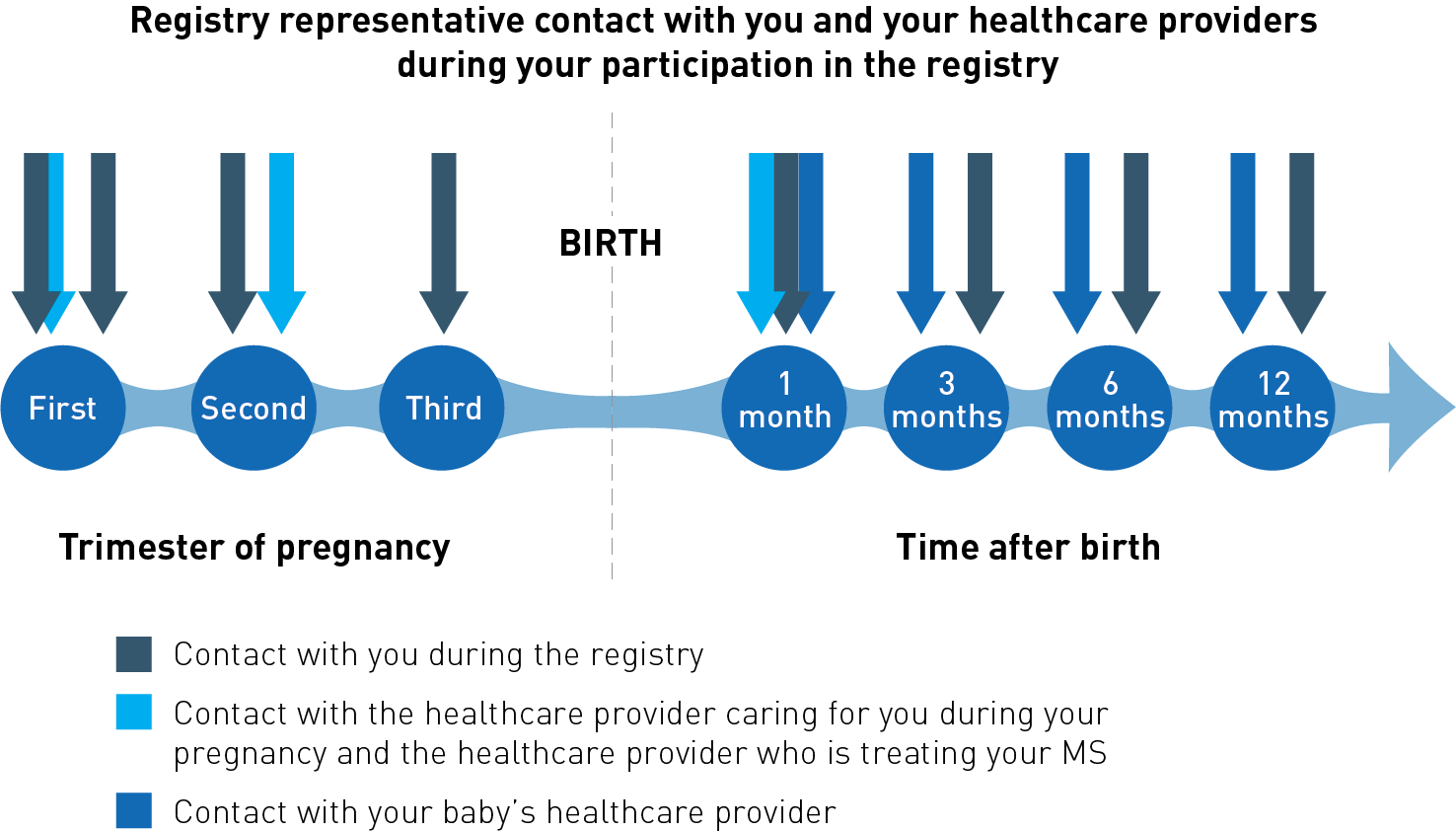
- call you once each trimester to ask if there have been any changes in your pregnancy, your MS and general health, the medicines you are taking, or any changes to your contact information
- call you about 4 weeks after you have given birth, and when your baby is approximately 3 months, 6 months, and 12 months old.
Why and when will my healthcare providers be contacted?
With your permission, a registry representative will contact the healthcare provider caring for you during your pregnancy, the healthcare provider who is treating your MS (your neurologist), and your baby’s healthcare provider, to collect information about your MS, your general health during pregnancy, and your baby’s health. Following your enrollment, a registry representative will:
- contact your healthcare providers, twice during your pregnancy, and about 4 weeks after you have given birth, to ask about any changes in your pregnancy, MS and general health, and treatment since the last call
- contact your baby’s healthcare provider about 4 weeks after you have given birth, and when your baby is 3, 6, and 12 months old to ask about the health of your baby.
The contact you and your healthcare providers will receive is summarized in the diagram below.
If you participate in the registry, you will not need to:
- attend any extra healthcare provider’s visits
- have any extra medical tests
- receive any additional medications.
What else will happen if I participate?
After you enroll in the registry, we will send you a welcome message by email. With your consent, we will also send you occasional emails and text messages. The texts and emails will remind you about upcoming telephone interviews. You can choose to stop receiving these communications at any time.
Our Privacy Policy contains full details of our commitment to your privacy.
Our FAQs contain answers to many frequently asked questions.
For the latest important safety information, please refer to the full Prescribing Information and Medication Guide. This is not intended to replace discussions with your healthcare provider.